PhD-project: Imaging the vesicular acetylcholine transporter with 18F-FEOBV PET in Parkinson’s disease
Jacob Horsager, October 2018
Main supervisor: Per Borghammer, Professor, MD, PhD, DMSc
Patients with Parkinson’s Disease (PD) often display symptoms from the gastrointestinal tract e.g. constipation due to loss of parasympathetic innervation. This cholinergic neuronal loss may be caused by α-synuclein aggregation, which has been hypothesized to start in the gut and spread via the vagus to the brain stem (and ultimately causing motor symptoms). This could explain why patients with PD often exhibit constipation up to 20 years before motor symptoms appear and why the vagus brainstem motor nucleus is one the first structures to be affected in PD. To achieve a detailed understanding of the early stages of PD, methods for visualizing and quantifying cholinergic denervation are needed.
The vesicular acetylcholine transporter (VAChT) is located almost solely in cholinergic presynaptic nerve terminals and are thus specific for cholinergic neurons. VAChT immunohistochemistry is considered gold standard for visualizing cholinergic nerve endings. Furthermore, studies have shown correlation between α-synuclein expression and the expression of VAChT, suggesting that cholinergic synapses may be particularly vulnerable in PD. 18F-fluoroethoxybenzovesamicol (18F-FEOBV) is a PET tracer that binds specifically to VAChT, and should be the ideal tracer to image parasympathetic denervation. Recently, the tracer was validated in a first-in-man study of healthy subjects, but the tracer is yet to be trialled in PD patients (Figure 1). The present PhD study will be the first to investigate the gastrointestinal tract in PD patients using 18F-FEOBV PET/CT and we believe it will yield detailed information of the early stages of this disease.
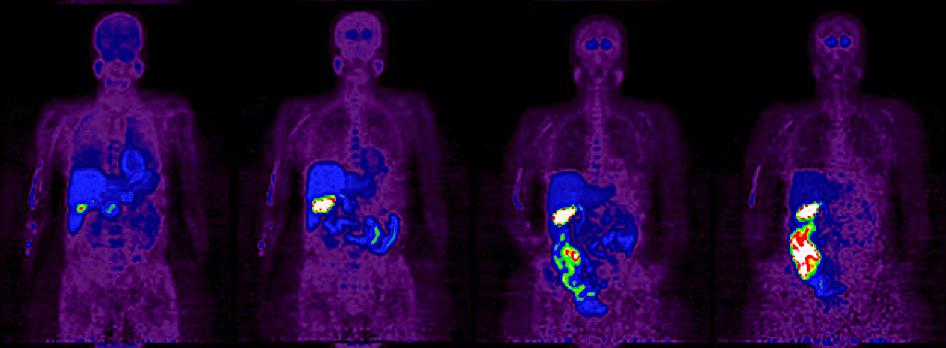
Figure 1. Whole-body 18F-FEOBV PET images of a healthy human subject. 50-min images obtained from 5, 60, 160, 360 min after tracer injection, respectively. (Petrou et al., JNM 2014)
We hypothesize that 18F-FEOBV is a specific marker of parasympathetic denervation. Compared to healthy subjects we hypothesize that PD patients will show decreased uptake of 18F-FEOBV in stomach, intestine, colon, pancreas, heart, and also in relevant regions of the brain. We expect the decreased tracer uptake in relevant organs to correlate with results from clinical tests of the autonomic system, i.e. delayed colonic transit time and increased orthostatic blood pressure drop. We hypothesize that PD patients have increased burden of symptoms from denervation of cholinergic neurons, such as constipation and bloating and that 18F-FEOBV uptake in relevant organs will correlate to these subjective symptoms evaluated by validated questionnaires.
In summary, the proposed PhD project is the first to study the human parasympathetic denervation of patients with PD using 18F-FEOBV PET/CT. It comprises a detailed evaluation of non-motor symptoms, which will contribute to give a comprehensive clinical interpretation of the PET/CT images. If successful, 18F-FEOBV PET/CT may have use as a clinical tool for investigating parasympathetic loss in early PD, which could be a valuable diagnostic marker in clinical trials. Furthermore, this PET method may find use in other disorders characterized by autonomic neuropathy, especially diabetes.