PhD project: The relationship between synaptic density and cognitive decline in Parkinson’s disease and early Dementia with Lewy Bodies
Katrine Andersen, October 2018
Main supervisor: Per Borghammer, Professor, MD, PhD, DMSc
Most patients with Parkinson’s disease (PD) develop cognitive decline and dementia during the course of the disease, but the underlying causes are poorly understood. In PD and Dementia with Lewy bodies (DLB) cognitive decline shows poor correlation with the defining pathological hallmark of these diseases, i.e. α-synuclein inclusions termed Lewy bodies and neurites. Similarly, in Alzheimer’s disease (AD), poor correlation is seen between amyloid plaque load and cognitive decline. However, in AD it has been shown that the loss of cortical synapses shows much stronger correlation with cognitive decline, as compared to levels of plaques and tangles.
A marked loss of cortical synapses has also been reported in DLB and PD patients with and without dementia, but correlations with cognitive decline remain unexplored. Thus, an improved understanding of synaptic loss in PD/DLB and its potential relation to cognitive decline and dementia is urgently needed, as this may lead to promising targets for new CNS drug therapies.
The novel PET tracer [11C]UCB-J binds to the presynaptic vesicle protein SV2A and has been validated as the first in vivo universal synaptic density marker in humans (Fig). The tracer is well tolerated and no adverse effects have been reported during more than 200 [11C]UCB-J PET scans in human subjects.
[11C]UCB-J PET showed decreased synaptic density in the temporal lobes, including hippocampus, of AD patients. A new study just demonstrated reduced synaptic density in the hippocampus of living people with AD by PET imaging of SV2A with this radioligand.
In patients with temporal lobe epilepsy, [11C]UCB-J PET demonstrated decreased synaptic density in the medial temporal lobe (Fig). Furthermore, decreased [11C]UCB-J binding was reported in the frontal cortex of patients with major depression, and global reduction of [11C]UCB-J binding was seen throughout the cortex in patients with chronic schizophrenia with the largest reduction in the amygdala. Thus, the ability of the tracer to quantify synaptic density is now well validated.
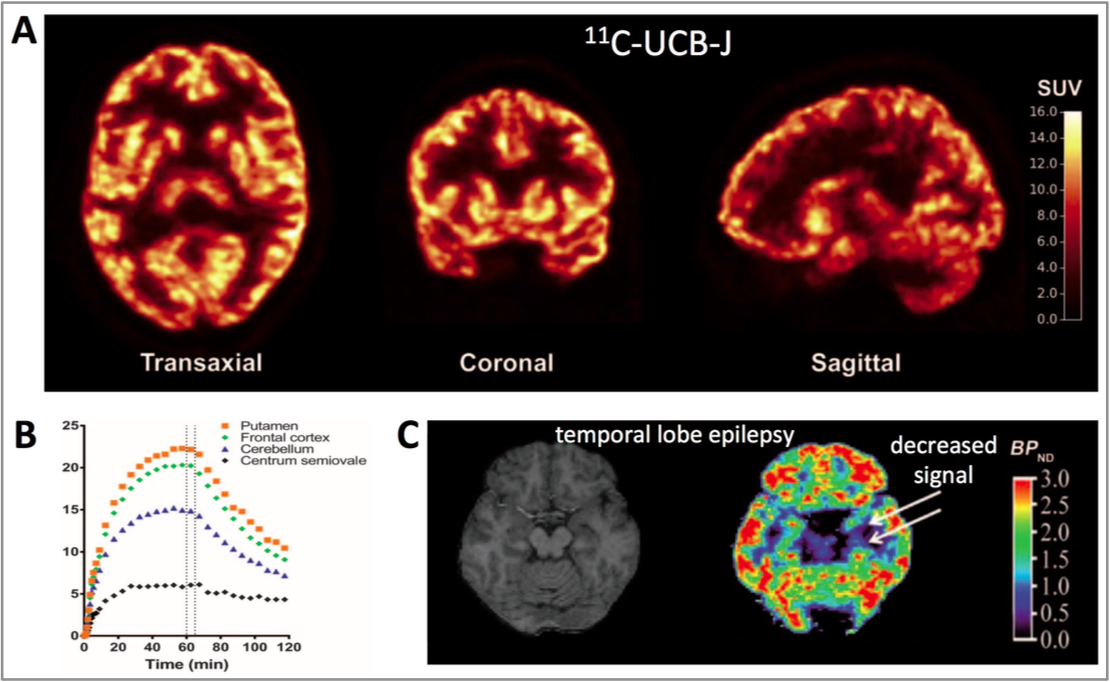
Figure. In humans, [11C]UCB-J exhibits excellent imaging properties. It shows very high levels of accumulation in grey matter with SUV values in the 9-15 range (Fig A). Also, more than 50% of the cortical tracer signal in grey matter but not in white matter (centrum semiovale) was displaced by the SV2A-specific ligand levetiracetam at 60 min (ventrical line), demonstrating the [11C]UCB-J binds with high specificity to SV2A (Fig B). A marked decrease of [11C]UCB-J PET binding was seen in the mesial temporal lobe of patients with temporal lobe epilepsy (Fig C).
We aim to study cortical synaptic integrity in PD & DLB patients. And to examine if the cortical synaptic density, measured by [11C]UCB-J PET, is correlated to cognitive decline and dementia, measured by neuropsychological test scores, and to expression of neuronal functional networks, measured by functional MRI scans.
With a diagnosis of PD and DLB follows a heavy burden of disability and-, adverse socioeconomic effects. Also, the impact on the patients and their families are devastating. These experiments may aid in making an early diagnosis, prognosticate, and elucidate the pathology behind these disorders. If our [11C]UCB-J study discloses important new knowledge about the pathophysiology of PD dementia, this will be an important break-through in our understanding of PD. This knowledge may open up a new field of scientific enquiry into synaptic dysfunction in PD, and lead to novel pharmaceutical interventions to treat or prevent cognitive decline in PD and DLB. [11C]UCB-J may therefore also have potential as a biomarker in future drug-trials.