MEG Research
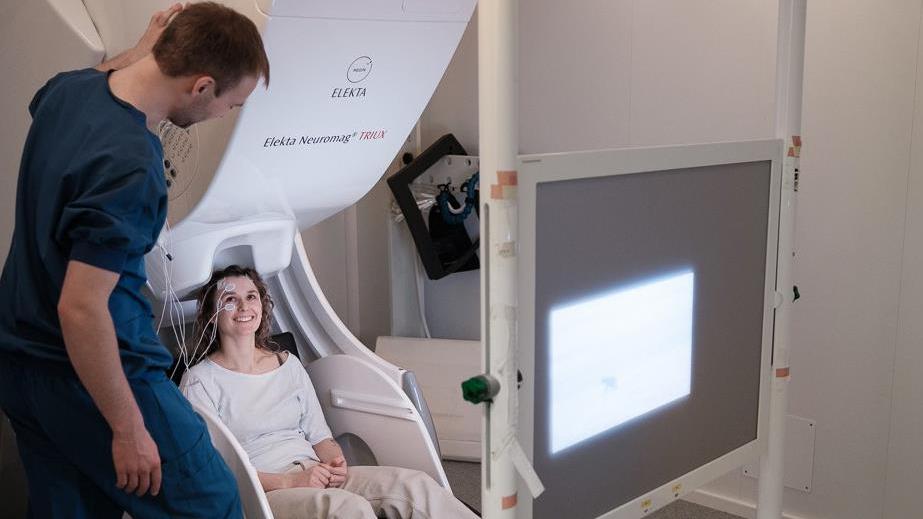
The Neuroradiology Research Unit is home to the first of Scandinavia’s two existing magnetoencephalography (MEG) systems. It measures the magnetic fields generated by the brain’s electrical activity. Unlike EEG signals, these magnetic fields are unaffected by the tissue surrounding the brain, offering unique localization and sensitivity.
The system is used by our colleagues from the Neurophysiology Clinic to improve the diagnostics and treatment as well as in a range of studies to understand brain function in health and disease, especially in terms of language functions and our visual system. Recently, we also acquired some of the first, next-generation biomagnetic sensors, which will further expand the utility of this research area.
Assessing language functions in health and disease
Developmental language deficits are estimated to affect ~10% of pre-school children; in adults, these are complemented by acquired deficits such as aphasia. Linguistic deficits also accompany a range of conditions such as autism and schizophrenia. In spite of the obvious importance of language and the high costs related to language deficits, it remains one of the least understood neurocognitive functions. One reason for this is that, as a communication system, human language is unparalleled in its complexity and has no suitable animal models.
Patients who are unable to cooperate with behavioural tests and follow instructions (aphasic or demented individuals, language-impaired children, etc.) are a challenge to clinicians assessing their linguistic ability; clear objective tools for assessment of neural language function without relying on overt behaviour are needed.
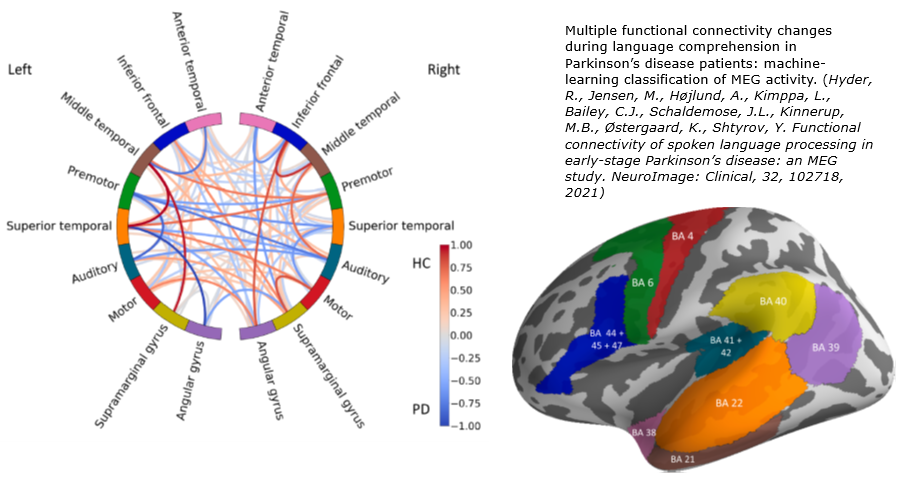
Using passive unattended speech presentation in EEG, MEG and fMRI, we have revealed automatic brain responses specific to different types of information (lexical, semantic, syntactic) that are independent of attention to the stimuli and do not require any overt behavioural responses from the subjects. This approach has a substantial potential as a means of assessing neurocognitive functions objectively and non-invasively, using e.g. patient-friendly MEG and/or inexpensive EEG available at most hospitals. We are working on improving these methodologies to optimize them as patient-friendly time-efficient protocols; these experiments include selecting sets of acoustically and psycholinguistically matched stimuli with robust and relevant linguistic contrasts, fine-tuning presentation modes to minimise testing time, streamlining analysis approaches.
Optically pumped magnetometers (OPMs)
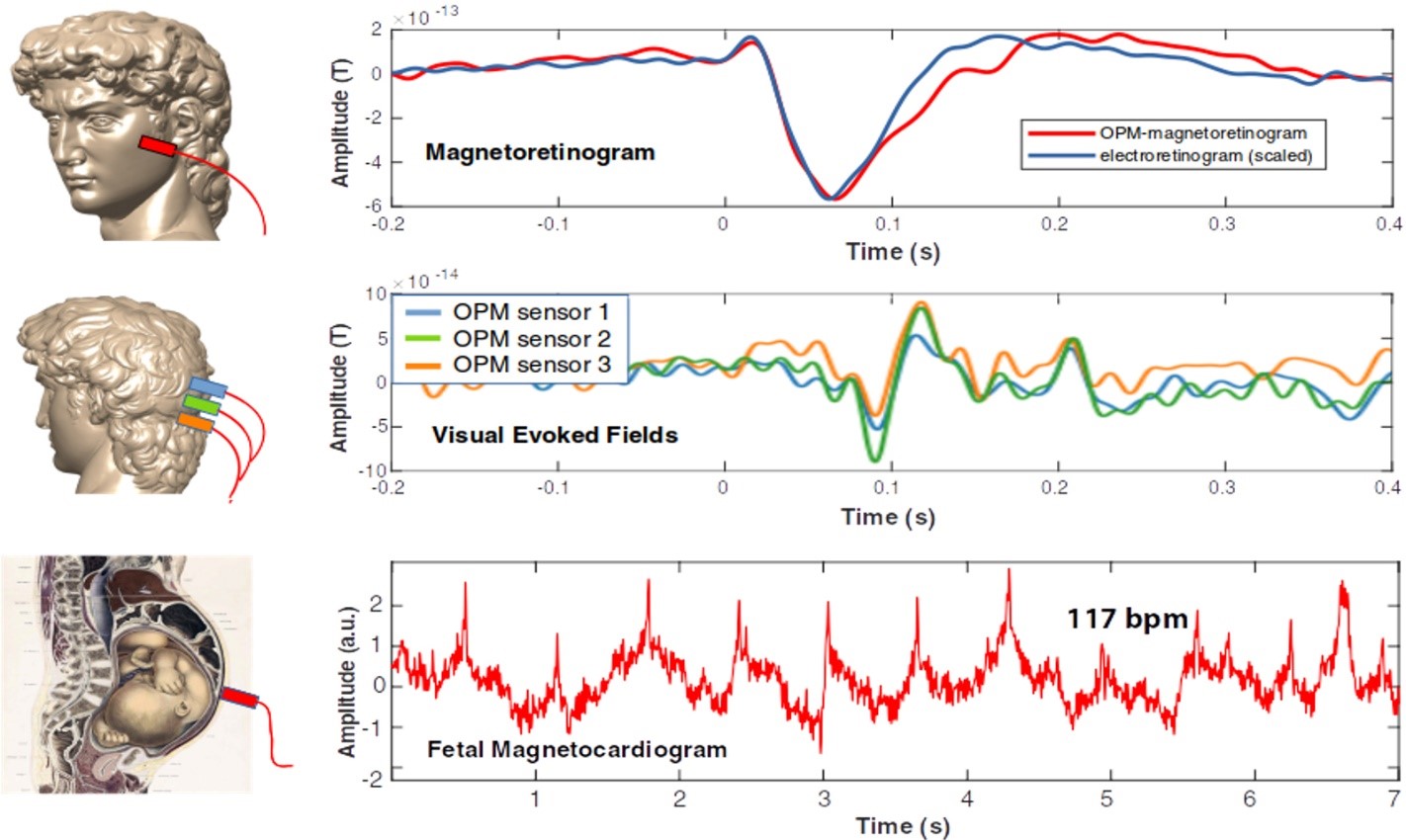
While traditional MEG depends on liquid Helium cooled Superconducting QUantum Interference Devices (SQUID), the next generation of magnetic field sensors, OPMs, operate at room temperature. Free of the liquid Helium dewar, sensors can therefore be placed close to biomagnetically active tissue, not only to the cerebral cortex. The figure shows pilot OPM recordings in our MEG laboratory. Upper row: Prof. Dalal was the first to demonstrate that OPMs can comfortably measure retinal responses from the cheek; a simultaneously recorded electroretinogram (with an electrode placed directly on the eye) is shown for comparison. Middle row: An OPM measurement of visual responses from visual cortex, with an OPM placed on the back of the head. Lower row: A fetal MCG from a healthy fetus at 33 weeks of gestation, with an OPM sensor placed on the mother’s abdomen.
Epilepsy detection
Using the MEG system, our colleagues at the neurophysiology clinic have found that patients suspected of epilepsy are better diagnosed using the power of MEG. When repeated awake and sleep EEG recordings (the current standard diagnostic approach) failed to detect abnormal brain activity patterns, a one hour combined EEG and MEG examination managed to detect evidence of epilepsy in 44% of the patients who were later diagnosed with epilepsy (Duez 2016). Indeed, in most of the cases where a diagnosis could be made, MEG alone could detect the critical, abnormal brain activity. In some epilepsy patients, seizures cannot be controlled by medicine, and surgical removal of the brain tissue causing their seizures may therefore be considered. The success of epilepsy surgery depends on the ability to localize the tissue in question. In epilepsy patients undergoing pre-surgical evaluation as part of the Danish Epilepsy Surgery Program, MEG and EEG data are used to localize the source of their seizures. These data provided new information that changed the course management of 34% of the patients (Duez 2019). In 80% of these patients, the information improved their treatment, pointing neurosurgeons to brain regions where existing methods had not been able to localize abnormalities, or to other regions than those deemed abnormal by other methods.
References
- Hyder R, Højlund A, Jensen M, Østergaard K, Shtyrov Y. Objective assessment of automatic language comprehension mechanisms in the brain: Novel E/MEG paradigm. Psychophysiology. 2020; 7(5):e13543. doi: 10.1111/psyp.13543.
- Gansonre C, Højlund A, Leminen A, Bailey C, Shtyrov Y.Task-free auditory EEG paradigm for probing multiple levels of speech processing in the brain. Psychophysiology. 2018;55(11):e13216. doi: 10.1111/psyp.13216.
- Andersen LM, Jerbi K, Dalal SS. Can EEG and MEG detect signals from the human cerebellum? Neuroimage. 2020 Apr 8;215:116817. doi: 10.1016/j.neuroimage.2020.116817.
- Leske S, Dalal SS. Reducing power line noise in EEG and MEG data via spectrum interpolation. Neuroimage. 2019 Apr 1;189:763-776. doi: 10.1016/j.neuroimage.2019.01.026.
- Sridharan KS, Højlund A, Johnsen EL, Sunde NA, Johansen LG, Beniczky S, Østergaard K. Differentiated effects of deep brain stimulation and medication on somatosensory processing in Parkinson's disease. Clin Neurophysiol. 2017;128(7):1327-1336. doi: 10.1016/j.clinph.2017.04.014.
- Duez L, Beniczky S, Tankisi H, Hansen PO, Sidenius P, Sabers A, Fuglsang-Frederiksen A. Added diagnostic value of magnetoencephalography (MEG) in patients suspected for epilepsy, where previous, extensive EEG workup was unrevealing. Clin Neurophysiol 127, 3301-3305 (2016).
- Duez L, Tankisi H, Hansen PO, Sidenius P, Sabers A, Pinborg LH, Fabricius M, Rásonyi G, Rubboli G, Pedersen B, Leffers AM, Uldall P, Jespersen B, Brennum J, Henriksen OM, Fuglsang-Frederiksen A, Beniczky S. Electromagnetic source imaging in presurgical workup of patients with epilepsy: A prospective study. Neurology 92, e576-e586 (2019).
Contacts
Professor Yury Shtyrov, e-mail: yury@cfin.au.dk
Professor Sarang S. Dalal, e-mail: sarang@cfin.au.dk
Professor Sándor Beniczky, e-mail: Sandor.Beniczky@aarhus.rm.dk
Neurophysiology clinic chair Peter Orm Hansen, e-mail: Peter.Orm.Hansen@rm.dk