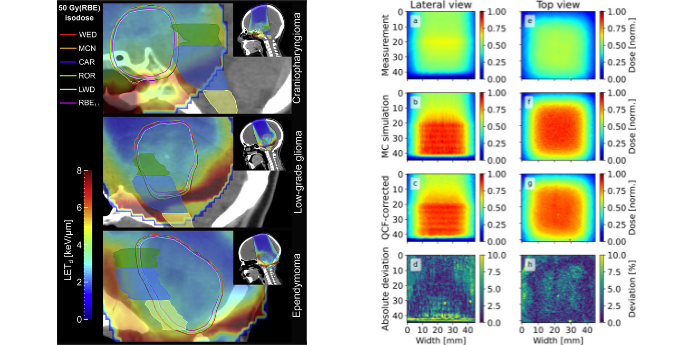
Image: Illustration of two of our research areas. Treatment plans of three children with brain tumours in different locations (left panel) showing the biologically equivalent dose contours (coloured lines) calculated using the clinical assumption that the effectiveness (known as the RBE) of protons per unit of radiation dose is the same as for conventional photon-based radiotherapy compared to that it is also varying with the linear energy transfer (LET) of the proton beam. Understanding the true biological effects of proton therapy is essential for its wide adoption. Several projects in our group are also developing solutions to measure in 3D the doses delivered with either proton therapy or advanced photon-based radiotherapy, using so-called radiochromic 3D dosimeters (right panel). These dosimeters are deformable and can be created in any shape (using 3D printing technology) and are currently helping us to verify delivery of proton therapy to tumours in the breast and in the liver, tumour sites influenced by breathing motion.
Our research group works on medical physics challenges close to the clinic. In our projects we work with clinical data – images, dose distributions and clinical outcomes from patients – with the aim of improving the treatment we can deliver to future patients. All projects in our group have project collaborators in the clinic (medical physicists and radiation oncologists), and several of our group members have part-time positions in the photon or proton clinic. We have a close collaboration with the Department of Physics and Astronomy at Aarhus University, and also work together with leading institutions in Europe and the US.
Most of our projects are focused on how proton therapy could be used in the best possible way, also for tumours in areas of the body influenced by anatomical changes. Many projects address challenges of how to make sure that the treatment the patient actually receive is as close as possible to what was planned, also for the most challenging patient groups. We are supporting the development of clinical proton therapy protocols and trials for prostate, paediatric, breast, head-and-neck and liver cancer.
The projects in our group can be categorised into three different themes. In the first area, we are investigating how we can improve the image-basis of treatment planning dose calculations for proton therapy. We are addressing computed-tomography (CT) imaging (including so-called dual energy CT) as well as online imaging modalities such as cone-beam CT; here we are working on approaches such as site-specific cone-beam calibrations, scatter-correction methods, and deep learning. These projects are tailored to improve our possibilities to calculate the 'dose-of-the-day' for our patients with head-and-neck, prostate and breast cancer, with perspectives such as dose-guidance, treatment adaptations and dose accumulations.
Several projects in our group are focusing on challenges in treatment planning and dose response modelling for proton therapy. In earlier as well as in on-going projects we have established the basis for the treatment planning strategy of the proton arm in a new multi-centre randomised trial for high-risk prostate cancer. Several of our projects deal with how to improve proton therapy for children with brain tumours, and how we can better predict and avoid unwanted side effects of their treatment. Many of the projects in this area are focused on understanding the role of what is known as the linear energy transfer (LET) of protons in determining better models for prediction of normal tissue effects after treatment.
In the third area we are working on tools to measure in 3D, and even 4D, the dose delivered to patients. We have developed a radiation detector in the form of a silicone-based material that is so-called radiochromic, i.e. it changes colour when it is irradiated. By reading out the colour change induced by radiation, we can measure the dose. Due to its silicone-base, the material is deformable. We are developing a patient 'model' (a so-called phantom) - with such dosimeter inserts - that we can deform similarly to what is happening in a patient during breathing. We are also studying how the dosimeter behaves when influenced by a magnetic field such as in combined units for magnetic resonance imaging and radiotherapy delivery (e.g. so-called MR-linacs). The projects in this area are supporting treatment protocols for breast cancer and liver tumours at our institution.
People
 Professor of medical physics
Professor of medical physics
Ludvig Muren
ludvmure@rm.dk
Further information.
Assistant Professor
Vicki Trier Taasti
vicki.taasti@rm.dk
Further information.
Specialty Registrar, postdoc
Stine Elleberg Petersen
stinpete@rm.dk
PURE, Aarhus University.
PhD student
Sofie Tilbæk Nielsen
SOTINI@rm.dk
PURE, Aarhus University.
 PhD student
PhD student
Casper Dueholm Vestergaard
Casper.Dueholm.Vestergaard@rm.dk
 PhD student
PhD student
Ivanka Sojat Tarp
Ivanka.Sojat@stab.rm.dk
PURE, Aarhus University. Further information.
PhD student
Kyriakos Fotiou
kyriakos.fotiou@rm.dk
 PhD student
PhD student
Lasse Bassermann
LASBAS@rm.dk
Collaborating researchers
 Medical Director, Professor
Medical Director, Professor
Morten Høyer
hoyer@aarhus.rm.dk
Further information.
 Professor of medical physics
Professor of medical physics
Per Rugaard Poulsen
per.poulsen@rm.dk
Further information.
 Consultant, associate professor of clinical medicine
Consultant, associate professor of clinical medicine
Kenneth Jensen
kenneth.jensen@auh.rm.dk
Further information.
Clinical Associate Professor, Dep. of Oncology, Aarhus University Hospital
Lise Nørgaard Bentzen
lise@oncology.au.dk. PURE, Aarhus University.
Senior Consultant
Yasmin Lassen
yasmin.lassen@auh.rm.dk
Medical Physicist
Jørgen Breede Baltzer Petersen
joerpete@rm.dk
PURE, Aarhus University. Further information.
Medical Physicist
Peter Sandegaard Skyt
peteskyt@rm.dk
Medical physicist, PhD
Ulrik Vindelev Elstrøm
ulrik.vindelev.elstroem@auh.rm.dk
PURE, Aarhus University.
Medical Physicist
Heidi Rønde
HEIDRE@rm.dk
Medical Physicist, PhD
Anne Vestergaard
annveste@rm.dk
Medical physicist
Maria Fuglsang Jensen
marfugje@rm.dk
Medical Physicist, PhD
Line Bjerregaard Stick
LINSTI@rm.dk
Medical Physicist
Liliana Stolarczyk
LILSTO@rm.dk
Medical physicist, Department of Oncology, Aarhus University Hospital
Steffen Hokland
Professor, Department of Physics and Astronomy, Aarhus University
Peter Balling
PURE, Aarhus University.
Assistant professor, Department of Physics and Astronomy, Aarhus University
Rosana Turtos
PURE, Aarhus University.
Associate professor, Department of Physics and Astronomy, Aarhus University
Brian Julsgaard
PURE, Aarhus University.